Our research
Our laboratory is interested in understanding the diverse signaling pathways and networks regulating essential basic cellular processes in normal and cancer cells. We use a multi-pronged approach in which science and technology from molecular biology, chemical biology, genetics, systems biology, immune-oncology, and novel mouse models of human cancer converge. In this way, we not only gain a better understanding of the alterations involved in tumor initiation, progression, metastasis and the immune microenvironment in tumors, but we also identify novel targets and develop new therapeutic regimens.
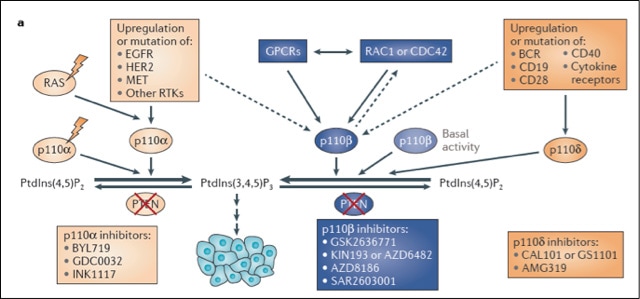
Discovery, determination and molecular understanding of divergent roles of the p110α and p110β isoforms of PI3K in cellular signaling, metabolism and tumorigenesis. Phosphatidylinositol 3-Kinases (PI3Ks) are a family of lipid kinases that play important roles in multiple physiological functions and cellular processes. Earlier studies on PI3K referred to this family of enzymes generically, as if they were a single entity. The Zhao laboratory, however, demonstrated that the two ubiquitously expressed PI3K isoforms, termed p110α and p110β, possess unique, non-redundant functions. In collaboration with Dr. Roberts, we developed conditional knockout mouse models for p110α and p110β, and pioneered a new front for understanding signal transduction by integrating mouse genetics and pharmacological approaches. Our work revealed previously unappreciated distinct roles for p110α and p110β in cellular signaling, metabolism and tumorigenesis, and changed the way we think of PI3K signaling. Moreover, these studies have laid a foundation for the new field of targeting specific PI3K isoforms in cancer, and guided the design of current trials of PI3K inhibitors in the clinic. Clearly, we still have much more to learn, such as the individual roles of these isoforms in mediating the immune microenvironment in tumors and the effects of their selective inhibition on immunotherapy.
Discovery, determination and molecular understanding of divergent roles of the p110α and p110β isoforms of PI3K in cellular signaling, metabolism and tumorigenesis. Phosphatidylinositol 3-Kinases (PI3Ks) are a family of lipid kinases that play important roles in multiple physiological functions and cellular processes. Earlier studies on PI3K referred to this family of enzymes generically, as if they were a single entity. The Zhao laboratory, however, demonstrated that the two ubiquitously expressed PI3K isoforms, termed p110α and p110β, possess unique, non-redundant functions. In collaboration with Dr. Roberts, we developed conditional knockout mouse models for p110α and p110β, and pioneered a new front for understanding signal transduction by integrating mouse genetics and pharmacological approaches. Our work revealed previously unappreciated distinct roles for p110α and p110β in cellular signaling, metabolism and tumorigenesis, and changed the way we think of PI3K signaling. Moreover, these studies have laid a foundation for the new field of targeting specific PI3K isoforms in cancer, and guided the design of current trials of PI3K inhibitors in the clinic. Clearly, we still have much more to learn, such as the individual roles of these isoforms in mediating the immune microenvironment in tumors and the effects of their selective inhibition on immunotherapy.
|
Adapted from Thorpe L, Yuzugullu H, Zhao JJ. PI3K in cancer: divergent roles of isoforms, modes of activation, and therapeutic targeting. Nature Rev. Cancer. 2015 Jan;15(1):7-24. PMCID: PMC4384662 |
Identification and therapeutic implication of the unique role PI3K-p110β that is critical in oncogenic signaling and tumorigenesis in the setting of PTEN-deficiency. PTEN loss is one of the most frequent events across all types of human cancer, and is associated with increased disease aggressiveness and poor outcome. We have been intensively investigating the role of p110β in PTEN-null tumors in several disease models, and identified its essential roles in prostate intraepithelial neoplasia (PIN), ovarian tumorigenesis, hematopoiesis and leukemogenesis, and PTEN hamartoma tumor syndrome (PHTS), a disorder caused by germline inactivating mutations in PTEN. Our findings in PHTS led to a joint patent between Dana-Farber and Novartis, and clinical trials are being designed to use PI3K inhibitors to treat patients with PHTS. In addition, my laboratory, in collaboration with Dr. Nathanael Gray at Dana-Farber, reported the first functional identification and characterization of a p110β-selective inhibitor, KIN193 (also known as AZD6482), with anti-tumor activity in mice. Our study suggests that this class of inhibitor holds great promise as a pharmacologic agent that could be used to address the potential therapeutic benefit of treating p110β-dependent PTEN-deficient human tumors. Currently, clinical trials with p110β-selective inhibitors are underway in patients with advanced PTEN-deficient solid tumors (NCT01458067 and NCT02465060) and in combination with anti-androgen therapy in metastatic castration-resistant prostate cancer (NCT02215096).
Creation of first-in-kind kinome-wide kinase libraries and identification of novel kinase targets in cancer. The Zhao laboratory has played a leading role in taking a systems biology approach to the study of kinase signaling in oncogenic transformation. We created first-in-kind “gain-of-function” kinase libraries, and have used these to identify novel oncogenic kinases, such as IKBKE and MAP3K8/COT1. More recently, using our in vivo transformation screen system, we identified a number of new kinase targets in breast cancer and astrocytomas, such as MELK, CDK7 and NTRK, hence providing opportunities for the development of novel therapies. In parallel, we have also taken kinome-wide “loss of function” approaches to search for tumor suppressor activity. We identified SIK1 as a novel kinase that couples LKB1 to p53-dependent anoikis and suppression of metastasis, thus establishing the LKB1-SIK1-p53 axis as a potentially important pathway in metastatic disease.
Overcoming therapeutic resistance: From mouse models to human therapy. Given that therapeutic resistance is a major obstacle in the clinic, we are particularly interested in identifying mechanisms of resistance to therapy. To this end, our novel GEMM of breast cancer driven by PIK3CAH1047R, coupled with pharmacological approaches, led to the discovery of a number of significant resistance mechanisms to PI3K-targeted therapy, such as spontaneous focal amplification of MET and MYC, and compensatory activation of the MAPK pathway. One of our most recent and exciting findings on this front is the identification of Cyclin D1-CDK4-mediated resistance to HER2-targeted therapy. Importantly, these findings led to the design of a randomized phase 2 clinical trial examining the role of CDK4/6 inhibitors in combination with HER2-targeted therapy as a regimen for patients with metastatic, refractory HER2+ breast cancer.
Over the past few years, we have been leading the effort at Dana-Farber to establish orthotopic patient-derived xenograft (PDX) models of metastatic breast cancer, with a focus on brain metastasis. In addition to being one of the hardest unmet needs in medical oncology, brain metastases are a particularly difficult scientific challenge, in large part due to the lack of clinically relevant models to study this disease. Our prominent work on establishing breast cancer brain metastasis (BCBM) PDX models has provided a remarkably strong system to investigate the molecular and genetic mechanisms that underlie metastasis and drug resistance in BCBM. Our pioneering work on this realm was published in Nature Medicine in July 2016 with a cover story and, more importantly, these initial findings are being translated into a phase 2 clinical trial. Following this lead, we are excited to continue working with Dana-Farber/Harvard Cancer Center (DF/HCC) oncologists to translate our fundamental preclinical findings into novel and improved therapeutic strategies for cancer patients.
Creation of first-in-kind kinome-wide kinase libraries and identification of novel kinase targets in cancer. The Zhao laboratory has played a leading role in taking a systems biology approach to the study of kinase signaling in oncogenic transformation. We created first-in-kind “gain-of-function” kinase libraries, and have used these to identify novel oncogenic kinases, such as IKBKE and MAP3K8/COT1. More recently, using our in vivo transformation screen system, we identified a number of new kinase targets in breast cancer and astrocytomas, such as MELK, CDK7 and NTRK, hence providing opportunities for the development of novel therapies. In parallel, we have also taken kinome-wide “loss of function” approaches to search for tumor suppressor activity. We identified SIK1 as a novel kinase that couples LKB1 to p53-dependent anoikis and suppression of metastasis, thus establishing the LKB1-SIK1-p53 axis as a potentially important pathway in metastatic disease.
Overcoming therapeutic resistance: From mouse models to human therapy. Given that therapeutic resistance is a major obstacle in the clinic, we are particularly interested in identifying mechanisms of resistance to therapy. To this end, our novel GEMM of breast cancer driven by PIK3CAH1047R, coupled with pharmacological approaches, led to the discovery of a number of significant resistance mechanisms to PI3K-targeted therapy, such as spontaneous focal amplification of MET and MYC, and compensatory activation of the MAPK pathway. One of our most recent and exciting findings on this front is the identification of Cyclin D1-CDK4-mediated resistance to HER2-targeted therapy. Importantly, these findings led to the design of a randomized phase 2 clinical trial examining the role of CDK4/6 inhibitors in combination with HER2-targeted therapy as a regimen for patients with metastatic, refractory HER2+ breast cancer.
Over the past few years, we have been leading the effort at Dana-Farber to establish orthotopic patient-derived xenograft (PDX) models of metastatic breast cancer, with a focus on brain metastasis. In addition to being one of the hardest unmet needs in medical oncology, brain metastases are a particularly difficult scientific challenge, in large part due to the lack of clinically relevant models to study this disease. Our prominent work on establishing breast cancer brain metastasis (BCBM) PDX models has provided a remarkably strong system to investigate the molecular and genetic mechanisms that underlie metastasis and drug resistance in BCBM. Our pioneering work on this realm was published in Nature Medicine in July 2016 with a cover story and, more importantly, these initial findings are being translated into a phase 2 clinical trial. Following this lead, we are excited to continue working with Dana-Farber/Harvard Cancer Center (DF/HCC) oncologists to translate our fundamental preclinical findings into novel and improved therapeutic strategies for cancer patients.